NIAAA Director's Report on Institute Activities to the 138th Meeting of the National Advisory Council on Alcohol Abuse and Alcoholism
Table of Contents
NIAAA BUDGET
NIAAA closed Fiscal Year (FY) 2014 on September 30th; the final appropriation for NIAAA was $446.3 million. A summary of key funding actions within this appropriation are as follows:
- NIAAA awarded 657 research project grants (RPGs), including 180 competing awards, which corresponds to a success rate of 19%.
- NIAAA funded 17 research centers for $26.4 million.
- NIAAA funded 137 other research grants for $36.1 million, including Career awards, one Cooperative Clinical Agreement, and several resource and conference grant awards.
- NIAAA supported 269 full-time training positions for $11.6 million.
- NIAAA funding for our Research and Development (R&D) contract portfolio was $36.5 million.
- NIAAA support for Intramural research totaled $49.0 million.
FY 2015
After a lengthy continuing resolution, HR 83, the Consolidated and Further Continuing Appropriations Act for FY 2015, was signed by the President on December 16, 2014. The omnibus appropriations bill is funding the entire government, except the Department of Homeland Security (DHS), through the end of FY 2015. DHS is being funded with a shorter-term continuing resolution through late February.
NIH received a total of $30.3 billion, $150 million above the FY 2014 level. This funding will continue basic bio-medical research and translational research through programs like the Clinical and Translational Science Awards (CTSA) and Institutional Development Award (IDeA) to help scientists discover cures. It includes specific increases for Alzheimer’s, cancer, and brain research, and $12.6 million for the Gabriella Miller Kids First Research Act pediatric research initiative.
The FY 2015 appropriation for NIAAA provides $447.2 million. This represents a $0.9 million or a 0.3% increase over the FY 2014 budget level. NIAAA estimates it will support a total of 654 RPGs in FY 2015, including 153 competing awards.
Of special note for NIAAA is language from the Joint Explanatory Statement of the Bill with a recommendation for NIAAA related to the health effects of moderate drinking. The following is an excerpt from this statement: “numerous epidemiological and basic science studies have demonstrated that moderate drinking can be beneficial to health by reducing risk for coronary artery disease, type 2 diabetes, and rheumatoid arthritis, among others. However, these studies used different protocols or questionnaires, and may be difficult to compare. The agreement encourages NIAAA to undertake a multicenter, multiyear clinical study to clarify the health impact of moderate alcohol consumption.” NIAAA will work with its partners to determine the best approach to respond to this request from Congress.
FY 2016
Preliminary work on the budget for FY 2016 is beginning. After intermediate stages of review, the President’s budget request will be presented to Congress in February 2015, at which time it will become available to the public.
NIAAA Budget (Dollars in Thousands)
|
FY 2014 ACTUAL |
FY 2015 ENACTED |
|||
| MECHANISM |
No. |
Amount |
No. |
Amount |
|---|---|---|---|---|
|
Research Projects |
||||
|
Noncompeting |
477 |
$184,107 |
501 |
$189,337 |
|
Administrative Supplements * |
(35) |
2,349 |
(24) |
1,269 |
|
Competing |
180 |
60,451 |
153 |
51,867 |
|
Subtotal, RPGs |
657 |
246,907 |
654 |
242,473 |
|
SBIR/STTR |
29 |
9,329 |
34 |
10,962 |
|
Research Project Grants |
686 |
256,236 |
688 |
253,435 |
|
Research Centers |
||||
|
Specialized/Comprehensive |
17 |
26,350 |
17 |
27,821 |
|
Clinical Research |
- |
- |
- |
- |
|
Biotechnology |
- |
- |
- |
- |
|
Comparative Medicine |
- |
- |
- |
- |
|
Res. Centers in Minority Instit. |
- |
- |
- |
- |
|
Subtotal, Centers |
17 |
26,350 |
17 |
27,821 |
|
Other Research |
||||
|
Research Careers |
93 |
14,304 |
93 |
13,834 |
|
Cancer Education |
- |
- |
- |
|
|
Cooperative Clinical Research |
1 |
6,750 |
1 |
7,180 |
|
Biomedical Research Support |
- |
- |
- |
|
|
Minority Biomed. Res. Support |
- |
- |
- |
|
|
Other |
43 |
15,091 |
46 |
14,254 |
|
Subtotal, Other Research |
137 |
35,145 |
140 |
35,268 |
|
Total Research Grants |
840 |
318,731 |
845 |
316,524 |
|
Training |
FTTP |
|||
|
Individual |
95 |
3,951 |
95 |
4,274 |
|
Institutional |
174 |
7,691 |
174 |
8,415 |
|
Total Training |
269 |
11,642 |
269 |
12,689 |
|
Research & Develop. Contracts |
68 |
36,762 |
65 |
38,000 |
|
(SBIR/STTR) * |
(8) |
(2,351) |
(4) |
(1,100) |
|
Intramural Research |
108 |
49,006 |
109 |
49,496 |
|
Res. Management & Support |
128 |
30,143 |
128 |
30,444 |
|
Total, NIAAA Budget Authority |
$446,284 |
$447,153 |
||
COLLABORATIVE RESEARCH ON ADDICTION AT NIH (CRAN) UPDATE
DIRECTOR’S ACTIVITIES
STAFF TRANSITIONS
 Dr. Jennifer A. Hobin joined the Office of Science Policy and Communications, Science Policy Branch in October 2014 as a Senior Health Science Policy Analyst. Dr. Hobin comes to NIAAA from the American Association for Cancer Research where she served as Director of Science Policy. Prior to AACR, Jennifer spent seven years at the Federation of American Societies for Experimental Biology where she worked on a broad range of research policy issues and played an instrumental role in the development of “myIDP,” a career planning tool for biomedical scientists with over 65,000 registered users. Jennifer earned her Ph.D. in biopsychology from the University of Michigan, Ann Arbor and was a Christine Mirzayan Science and Technology Policy Graduate Fellow at the National Academies.
Dr. Jennifer A. Hobin joined the Office of Science Policy and Communications, Science Policy Branch in October 2014 as a Senior Health Science Policy Analyst. Dr. Hobin comes to NIAAA from the American Association for Cancer Research where she served as Director of Science Policy. Prior to AACR, Jennifer spent seven years at the Federation of American Societies for Experimental Biology where she worked on a broad range of research policy issues and played an instrumental role in the development of “myIDP,” a career planning tool for biomedical scientists with over 65,000 registered users. Jennifer earned her Ph.D. in biopsychology from the University of Michigan, Ann Arbor and was a Christine Mirzayan Science and Technology Policy Graduate Fellow at the National Academies. Dr. Falk Lohoff recently joined the Laboratory of Clinical & Translational Studies, Division of Intramural Clinical and Biological Research, as Chief of the Section on Clinical Genomics and Experimental Therapeutics. Dr. Lohoff is a Lasker Clinical Research Scholar at NIH. He received his medical degree from Humboldt University of Berlin in 2002, and completed residency training in psychiatry and a fellowship in neuropsychopharmacology at the University of Pennsylvania. He is board certified in Psychiatry since 2007. He was Assistant Professor of Psychiatry at the University of Pennsylvania from 2007-2014, after which he joined the NIH intramural program as a Lasker Clinical Research Scholar. Dr. Lohoff has worked on clinical trials in mood and anxiety disorders for the last seven years and was also involved in direct patient care at the University of Pennsylvania. His research is focused on translational medicine and spans areas of molecular genetics, epigenetics, imaging-genetics, pharmacogenetics, and clinical experimental trials.
Dr. Falk Lohoff recently joined the Laboratory of Clinical & Translational Studies, Division of Intramural Clinical and Biological Research, as Chief of the Section on Clinical Genomics and Experimental Therapeutics. Dr. Lohoff is a Lasker Clinical Research Scholar at NIH. He received his medical degree from Humboldt University of Berlin in 2002, and completed residency training in psychiatry and a fellowship in neuropsychopharmacology at the University of Pennsylvania. He is board certified in Psychiatry since 2007. He was Assistant Professor of Psychiatry at the University of Pennsylvania from 2007-2014, after which he joined the NIH intramural program as a Lasker Clinical Research Scholar. Dr. Lohoff has worked on clinical trials in mood and anxiety disorders for the last seven years and was also involved in direct patient care at the University of Pennsylvania. His research is focused on translational medicine and spans areas of molecular genetics, epigenetics, imaging-genetics, pharmacogenetics, and clinical experimental trials. Erin Manor was selected to fill the position of Chief, Administrative Services Branch. Previously, Erin served as the Intramural Research Program Administrative Services Section Chief at NIAAA. Prior to NIAAA, Erin was a Supervisory Administrative Officer with NIMH, and also worked with the Intramural Research Program of NIDA in Baltimore.
Erin Manor was selected to fill the position of Chief, Administrative Services Branch. Previously, Erin served as the Intramural Research Program Administrative Services Section Chief at NIAAA. Prior to NIAAA, Erin was a Supervisory Administrative Officer with NIMH, and also worked with the Intramural Research Program of NIDA in Baltimore. Joan Romaine, M.P.H., joined the Office of the Director in October 2014, as a Health Specialist working under Dr. Kendall Bryant in the HIV/AIDS Research Program. Joan’s background is in public health, with a focus on international health policy and programs. She has been working on HIV/AIDS since 1995 when she served as a Peace Corps Volunteer in Gabon’s National AIDS Program in Libreville, Gabon. Joan comes to NIAAA from the NIH Office of AIDS Research (OAR) where she spent three years working on OAR-supported HIV/AIDS bilaterals with Russia and South Africa, as well as overseeing the Program Planning and Analysis group, which is responsible for the coordination of the development of the Trans-NIH Plan for HIV-Related Research and OAR’s Advisory Council. Prior to that, Joan served on a detail to the White House Office of National AIDS Policy where she helped on activities and reports related to the implementation of the National HIV/AIDS Strategy. Before going to the White House, Joan worked in the Division of AIDS at NIAID for nine years, serving as the Institute’s AIDS Coordinator for three years as well as working on scientific communications and initiatives, including the Comprehensive International Program of Research on AIDS (CIPRA). Joan began at NIH in 2001 as a Presidential Management Intern working at NIAAA on the College Drinking Prevention initiative.
Joan Romaine, M.P.H., joined the Office of the Director in October 2014, as a Health Specialist working under Dr. Kendall Bryant in the HIV/AIDS Research Program. Joan’s background is in public health, with a focus on international health policy and programs. She has been working on HIV/AIDS since 1995 when she served as a Peace Corps Volunteer in Gabon’s National AIDS Program in Libreville, Gabon. Joan comes to NIAAA from the NIH Office of AIDS Research (OAR) where she spent three years working on OAR-supported HIV/AIDS bilaterals with Russia and South Africa, as well as overseeing the Program Planning and Analysis group, which is responsible for the coordination of the development of the Trans-NIH Plan for HIV-Related Research and OAR’s Advisory Council. Prior to that, Joan served on a detail to the White House Office of National AIDS Policy where she helped on activities and reports related to the implementation of the National HIV/AIDS Strategy. Before going to the White House, Joan worked in the Division of AIDS at NIAID for nine years, serving as the Institute’s AIDS Coordinator for three years as well as working on scientific communications and initiatives, including the Comprehensive International Program of Research on AIDS (CIPRA). Joan began at NIH in 2001 as a Presidential Management Intern working at NIAAA on the College Drinking Prevention initiative.HONORS AND AWARDS
Dr. George Koob received the 2014 Innovator Award from the Treatment Research Institute in recognition of his scientific contributions and outstanding leadership in advancing understanding of how the brain and body respond to alcohol consumption.
Dr. Kari Johnson, postdoctoral fellow in the Section on Synaptic Pharmacology, Laboratory for Integrative Neuroscience, was awarded Postdoctoral Research Associate (PRAT) fellowship. The award was effective October 1, 2014. This is a prestigious fellowship given to outstanding trainees for training in pharmacological sciences.
Dr. Julia Lemos, PRAT fellow in the Section on Neuronal Structure, Laboratory for Integrative Neuroscience, received a travel fellowship to attend the annual meeting of the American College of Neuropsychopharmacology. The meeting took place December 7-11, 2014.
Five NIH postdoctoral fellows received Fellows Awards for Research Excellence for the 2015 fiscal year. This competitive and prestigious award can be used to support travel to national or international meetings where the fellow is presenting their research. The winners are: Dr. Ozge Gunduz Cinar, Research Fellow in the Laboratory of Behavioral and Genomic Research; Dr. Hadley Bergstrom, Intramural Research Training Fellow in the Laboratory of Behavioral and Genomic Research; Dr. Nicholas Jury, Intramural Research Training Fellow in the Laboratory of Behavioral and Genomic Research; Dr. Dechun Feng, Visiting Fellow in the Laboratory of Liver Diseases; and Dr. Lauren Dobbs, Intramural Research Training Fellow in the Section on Research Structure, Laboratory for Integrative Neuroscience.
Dr. Carlos Cortes, Intramural Research Training Fellow in the Section on Clinical Psychoneuroendocrinology and Neuropsychopharmacology, Laboratory of Clinical & Translational Studies, received a travel fellowship to attend the 2015 annual meeting of the Society of Biological Psychiatry. The meeting will take place May 14-16, 2015.
Dr. Tony Jourdan, Visiting Fellow in the Section on Neuroendocrinology, Laboratory of Physiological Studies, received a travel fellowship to attend a New York Academy of Sciences conference on Diabetic Kidney Disease. The meeting took place December 9, 2014.
NEW REQUEST FOR APPLICATIONS (RFAS) AND PROGRAM ANNOUNCEMENTS (PA’S)
The following Notice of Funding Opportunities (NOFOs) were issued by NIAAA:
RFA-AA-15-007: Alcohol Biosensors (STTR)(R41/R42). The purpose of this NOFO is to solicit Small Business Technology Transfer (STTR) applications from eligible small business concerns proposing to design and produce a wearable device to monitor blood alcohol levels in real time.
RFA-AA-15-008: Alcohol Biosensors (SBIR)(R43/R44). The purpose of this NOFO is to solicit Small Business Innovation Research (SBIR) applications from eligible small business concerns proposing to design and produce a wearable device to monitor blood alcohol levels in real time.
RFA-AA-15-004: Consortium on the Neurobiology of Adolescent Drinking in Adulthood (NADIA) Research Projects (U01). The purpose of this NOFO is to support collaborative research projects (U01) focusing on research efforts across different research institutions investigating the consequences of repeated adolescent alcohol exposure on brain maturation and adult abilities.
RFA-AA-15-005: Consortium on the Neurobiology of Adolescent Drinking in Adulthood (NADIA) Administrative Resource (U24). This NOFO solicits Administrative Resources-Cooperative Agreements (U24) to provide administrative support to the research projects (U01) as part of the Consortium on the Neurobiology of Adolescent Drinking in Adulthood (NADIA).
RFA-AA-15-006: Consortium on the Neurobiology of Adolescent Drinking in Adulthood (NADIA) Research Resource (U24). This NOFO solicits Research Resources-Cooperative Agreements (U24) to provide scientific (core) support to the research projects (U01) as part of the Consortium on the Neurobiology of Adolescent Drinking in Adulthood (NADIA).
PAR-15-073: Investigational New Drug (IND)-enabling Development of Medications to Treat Alcohol Use Disorder and Alcohol-related Disorders (UT2). This NOFO directed to Small Business Technology Transfer (STTR) applicants is dedicated to the development of therapeutic agents for disorders that fall under the mission of NIAAA.
PAR-15-074: Investigational New Drug (IND)-enabling Development of Medications to Treat Alcohol Use Disorder and Alcohol-related Disorders (U44). This NOFO encourages Small Business Innovation Research (SBIR) grant applications from small business concerns (SBCs) that propose the development of therapeutic agents for disorders that fall under the mission of NIAAA.
PAR-15-054: Alcohol Education Project Grants (R25). The over-arching goal of the NIAAA R25 program is to foster a better understanding of biomedical, behavioral and clinical research and its implications. To accomplish the stated over-arching goal, this NOFO will support creative educational activities with a primary focus on Curriculum or Methods Development and Outreach activities for Health Professionals.
PA-15-026: Mechanistic Studies of Pain and Alcohol Dependence (R01). This NOFO encourages applications that propose to conduct mechanistic studies on the relationship between excessive alcohol drinking, alcohol dependence, and pain.
PAR-14-338: Secondary Analyses of Existing Alcohol Epidemiology Data (R01). This NOFO encourages the submission of investigator-initiated research grant applications to support the secondary analysis of existing data sets with the goal of enhancing our understanding of patterns of alcohol consumption and the epidemiology of alcohol-related problems.
PA-14-337: Secondary Analyses of Existing Alcohol Epidemiology Data (R03). This NOFO encourages the submission of investigator-initiated research grant applications to support the secondary analysis of existing data sets with the goal of enhancing our understanding of patterns of alcohol consumption and the epidemiology of alcohol-related problems.
PA-14-336: Secondary Analyses of Existing Alcohol Epidemiology Data (R21). This NOFO encourages the submission of investigator-initiated research grant applications to support the secondary analysis of existing data sets with the goal of enhancing our understanding of patterns of alcohol consumption and the epidemiology of alcohol-related problems.
PA-15-058: Unconventional Roles of Ethanol Metabolizing Enzymes, Metabolites, and Cofactors in Health and Disease (R01). The purpose of this NOFO is to provide support for integrated, innovative research on the novel and unconventional contributions of ethanol metabolizing pathways, their metabolites, cofactors, and interactions with synergizing biological pathways in the development of alcohol-induced diseases and end organ injuries.
PA-15-057: Unconventional Roles of Ethanol Metabolizing Enzymes, Metabolites, and Cofactors in Health and Disease (R21). The purpose of this NOFO is to provide support for integrated, innovative research on the novel and unconventional contributions of ethanol metabolizing pathways, their metabolites, cofactors, and interactions with synergizing biological pathways in the development of alcohol-induced diseases and end organ injuries.
NIAAA is participating in the following NOFOs:
RFA-NS-15-003:BRAIN Initiative: New Technologies and Novel Approaches for Large-Scale Recording and Modulation in the Nervous System (U01). This NOFO seeks applications for proof-of-concept testing and development of new technologies and novel approaches for large scale recording and manipulation of neural activity, to enable transformative understanding of dynamic signaling in the nervous system.
RFA-NS-15-004:BRAIN Initiative: Optimization of Transformative Technologies for Large Scale Recording and Modulation in the Nervous System (U01).This NOFO seeks applications for the optimization of existing and emerging technologies and approaches that have potential to address major challenges associated with recording and manipulating neural activity, at or near cellular resolution, at multiple spatial and temporal scales, in any region and throughout the entire depth of the brain.
RFA-NS-15-005:BRAIN Initiative: Integrated Approaches to Understanding Circuit Function in the Nervous System (U01).The purpose of this NOFO is to promote the integration of experimental, analytic, and theoretical capabilities for large-scale analysis of neural systems and circuits. This NOFO seeks applications for exploratory research studies that use new and emerging methods for large scale recording and manipulation of neural circuits across multiple brain regions. Applications should propose to elucidate the contributions of dynamic circuit activity to a specific behavioral or neural system.
RFA-MH-15-200: BRAIN Initiative: Planning for Next Generation Human Brain Imaging (R24) (U01).This NOFO, in support of the NIH Brain Research through Advancing Innovative Neurotechnologies (BRAIN) Initiative, aims to support planning activities and the initial stages of development of entirely new or next generation brain imaging technologies and methods that will lead to transformative advances in our understanding of the human brain.
RFA-LM-15-002: NIH Big Data to Knowledge (BD2K) Initiative Research Education: Open Educational Resources for Sharing, Annotating and Curating Biomedical Big Data (R25). The NIH Research Education Program (R25) supports research education activities in the mission areas of the NIH. The over-arching goal of this BD2K R25 funding announcement is to complement and/or enhance the training of a workforce to meet the nation’s biomedical, behavioral, and clinical research needs. To accomplish the stated over-arching goal, this NOFO will focus on Curriculum or Methods Development. In particular, this NOFO seeks applications for development of open educational resources.
RFA-LM-15-001: NIH Big Data to Knowledge (BD2K) Initiative Research Education: Massive Open Online Course (MOOC) on Data Management for Biomedical Big Data (R25). The NIH Research Education Program (R25) supports research education activities in the mission areas of the NIH. The over-arching goal of this BD2K R25 NOFO is to complement and/or enhance the training of a workforce to meet the nation’s biomedical, behavioral, and clinical research needs. To accomplish the stated over-arching goal, this NOFO will focus on Curriculum or Methods Development. In particular, this NOFO seeks applications for development of an open, online educational resource.
RFA-HD-15-029: Pediatric HIV/AIDS Cohort Study (PHACS) Data and Operations Center (DOC) (U01). The purpose of this NOFO is to continue support for the Pediatric HIV/AIDS Cohort Study (PHACS) which addresses critical scientific questions on the clinical course of perinatally acquired HIV infection in adolescents and young adults and the oral and systemic health consequences of in utero and infant exposure to antiretroviral chemotherapy in representative cohorts of children in the United States.
PAR-15-047: Systems Science and Health in the Behavioral and Social Sciences (R21). This NOFO is intended to increase the breadth and scope of topics that can be addressed with systems science methodologies. This NOFO calls for research projects that are applied and/or basic in nature (including methodological and measurement development), have a human behavioral and/or social science focus, and employ methodologies suited to addressing the complexity inherent in behavioral and social phenomena, referred to as systems science methodologies. Additionally, this NOFO seeks to promote interdisciplinary collaboration among health researchers and experts in computational approaches to further the development of modeling- and simulation-based systems science methodologies and their application to important public health challenges.
PAR-15-048: Systems Science and Health in the Behavioral and Social Sciences (R01). This NOFO is intended to increase the breadth and scope of topics that can be addressed with systems science methodologies. This NOFO calls for research projects that are applied and/or basic in nature (including methodological and measurement development), have a human behavioral and/or social science focus, and employ methodologies suited to addressing the complexity inherent in behavioral and social phenomena, referred to as systems science methodologies. Additionally, this NOFO seeks to promote interdisciplinary collaboration among health researchers and experts in computational approaches to further the development of modeling- and simulation-based systems science methodologies and their application to important public health challenges.
PA-15-046: Administrative Supplements for Tobacco Regulatory Research on the Public Display of Harmful and Potentially Harmful Constituents (HPHC) Information (Admin Supp). The purpose of this NOFO is to generate data to support the implementation of public displays of HPHC information.
PA-15-035: Research Aimed at Novel Behavioral Targets to Improve Adolescent Substance Abuse Treatment and Prevention Interventions (R34). This NOFO is part of the CRAN initiative. Areas supported by this NOFO include research to inform the generation and refinement of novel targets for substance abuse treatment and prevention interventions, modules or adjuncts to existing treatments and prevention interventions that seek to target and modulate behavioral or neurobehavioral processes (e.g., impulsivity, risk-taking propensity, sensation and novelty seeking, distress tolerance, delay discounting, self-regulation, stress reactivity) in adolescents.
PA-15-034: Administrative Supplements for Research on Sex/Gender Differences (Admin Supp). The Office of Research on Women's Health (ORWH) announces the availability of administrative supplements to support research highlighting the impact of sex/gender differences (or similarities) and/or sex and gender factors in human health and illness, including basic, preclinical, clinical, and behavioral studies
NOTABLE NIAAA STAFF ACTIVITIES
Dr. Dale Hereld co-authored The Safe Passage Study: Design, Methods, Recruitment, and Follow-Up Approach (Dukes et al., 2014), which appeared in Paediatric and Perinatal Epidemiology in September 2014. This publication describes the study design, hypotheses, and methodologies employed by the NIAAA/NICHD/NIDCD-supported Prenatal Alcohol and SIDS and Stillbirth (PASS) Network to assess the role of alcohol in adverse birth outcomes including sudden infant death syndrome (SIDS) and stillbirth. As a Project Scientist, Dr. Hereld represents NIAAA in this effort.
Drs. Svetlana Radaeva and Gary Murray from NIAAA and Dr. Gyongyi Szabo from the University of Massachusetts organized a special closed session meeting of the principal investigators for the Alcoholic Hepatitis consortia on November 11, 2014 in Boston in conjunction with the American Association for the Study of Liver Diseases meeting. Dr. George Koob chaired the meeting, provided comments on the progress of the various consortia, and encouraged the formation of a steering committee to guide the progression of the independent consortia toward a network of consortia with a shared mission and enhanced cooperation.
Dr. Robert B. Huebner delivered a talk titled, “Screening and Brief Interventions across Settings, Patients, Patient Populations, and Providers” at the 11th Congress of the International Network on Brief Interventions for Alcohol and Other Drugs. The conference was held at the Medical University of Warsaw in Warsaw, Poland on September 18–19, 2014.
Dr. Deidra Roach gave a presentation titled “Women and Alcohol” at the first annual “Women in Recovery Conference” sponsored by Addiction Treatment Services in Traverse City, Michigan on November 14, 2014. This talk focused on recent trends in drinking among girls and women, the effects of excessive drinking on girls and women over the lifespan, and promising approaches to treatment and recovery support.
Dr. Lori Ducharme delivered a plenary talk titled “Scaling Up, Scaling Out, Scaling Back: Implementation Priorities in Addiction Health Services Research” at the annual Addiction Health Services Research Conference at Boston University, on October 16, 2014. This presentation discussed the need for research to support the broad uptake of evidence-based practices for alcohol and drug treatment services.
Dr. Antonio Noronha was invited to be a discussion participant in two symposia at the European Society for Biomedical Research on Alcoholism Nordmann Award Meeting in Sofia, Bulgaria, September 20-22, 2014.
Dr. Ralph Hingson gave a talk titled “Preventing Drug-Impaired Driving: Lessons from Alcohol-Impaired Driving” at the World Health Organization’s Technical Consultation on Drug Use And Road Safety meeting in Geneva, Switzerland, on December 18, 2014. This talk discussed lessons learned from the experience of the United States in reducing alcohol-impaired driving and recommended ways other countries could apply these lessons to help prevent drug-impaired driving.
Dr. Ralph Hingson gave a talk titled “New Research since the Surgeon General’s Call to Action to Prevent and Reduce Underage Drinking” at the Coalition for Healthy Youth’s Prevention of Youth Substance Abuse in Rural Communities Conference in Lancaster, South Carolina, on August 6, 2014. This talk outlined new research on trends in and consequences of underage drinking and interventions to prevent and reduce underage drinking that have emerged since the 2007 Call to Action.
Dr. Ralph Hingson gave two talks at the eighth annual meeting of the Georgia School of Addiction Studies on August 25, 2014. Dr. Hingson’s first presentation was “New Research since the Surgeon General’s Call to Action to Prevent and Reduce Underage Drinking.” This talk outlined new research on trends in and consequences of underage drinking, and discussed interventions to prevent and reduce underage drinking that have emerged since the 2007 Call to Action. His second talk was “Preventing Alcohol-Related Injury: From Global to Local.” This talk addressed how to best prevent alcohol-related injury at the global, national, state, and community levels.
Dr. Ralph Hingson gave a talk titled “Preventing Alcohol-Related Injury” at the University of Massachusetts Amherst’s School of Public Health and Health Science’s meeting on The Complexity of Alcohol Use: The Role of Science, Government, and the Community in Amherst, Massachusetts on October 29, 2014. This talk addressed how to best prevent alcohol-related injury, especially at the state and community levels.
Drs. Kenneth Warren and Peggy Murray presented at the First Baltic Conference on the Prevention of Fetal Alcohol Spectrum Disorders in Vilnius Lithuania on September 26, 2014. Dr. Warren gave the keynote presentation “Alcohol and Pregnancy in History and Art: Missed Clues along the Way.” Dr. Murray gave a plenary talk, “Alcohol Policy Interventions to Prevent Fetal Alcohol Spectrum Disorders.” On this trip, Drs. Warren and Murray also visited with Dr. Eva Tomberg, head of children’s welfare programs for the Department of Health in Tallinn, Estonia to discuss joint issues in the treatment and prevention of fetal alcohol spectrum disorders (FASD) and maternal drinking during pregnancy.
Drs. Kenneth Warren and Peggy Murray both gave presentations at the Third European Conference on Fetal Alcohol Spectrum Disorders in Rome on October 20, 2014. Dr. Warren gave the opening keynote presentation titled “Alcohol and Pregnancy in History and Art: Missed Clues along the Way." Dr. Murray presented a plenary talk titled, “Joining Forces: Using Global Health Policy to Address Fetal Alcohol Spectrum Disorders in Europe and North America,” on October 22, 2014.
Dr. Kenneth Warren was the keynote speaker at the 25th Anniversary Celebration of the Center on Alcoholism, Substance Abuse, and Addictions (CASAA) at the University of New Mexico on November 7, 2014. Dr. Warren addressed the research advances that have emerged over the past 25 years and the contributions that CASAA investigators have made in this period in advancing research on alcohol abuse and alcoholism
WHAT’S AHEAD?
Dr. Andras Orosz of the Division of Metabolism and Health Effects has proposed a new conference series: “Alcohol-Induced End Organ Diseases” that has been recently approved by the Gordon Research Conference Evaluation Committee and Board of Trustees and is scheduled for early 2017. Conferences will be held every second year and will provide an important forum for discussions of the tissue and organ damage caused by excessive alcohol use.
In May 2015, NIAAA’s Division of Treatment and Recovery Research will sponsor a research track at the American Psychiatric Association meeting in Toronto, Canada titled “Advances in Treatment of Alcohol and Co-occurring Psychiatric Disorders across Patient Populations and Settings.” It will focus on how alcohol research can inform and improve the treatment of co-occurring alcohol use and psychiatric disorders across patient populations and settings. Treating co-occurring alcohol use and psychiatric disorders is complex and our research track reflects that complexity. We will host presentations on the underlying neurobiology of alcohol use disorders, new trends in pharmacotherapies, PTSD, binge drinking among youth, fetal alcohol syndrome, and the use of both alcohol and marijuana by young people
NIAAA RESEARCH HIGHLIGHTS
Significance: Higher levels of maternal alcohol consumption during pregnancy were associated with lower levels of axial diffusivity (a measure of axonal structural integrity) in white matter fiber tracts in the brain of two-week-old human newborns while levels of radial diffusivity (a measure of myelination) were not. This study suggests that prenatal alcohol exposure has effects primarily on axonal structure which could impair axonal growth and guidance during this critical period of brain development. The study also demonstrates the valuable information about brain structure that can now be gained from even newborn infants with neuroimaging techniques.
Prenatal alcohol exposure (PAE) is known to have severe, long-term consequences for brain and behavioral development already detectable in infancy and childhood. Resulting features of fetal alcohol spectrum disorders include cognitive and behavioral effects, as well as facial anomalies and growth deficits. Diffusion tensor imaging (DTI) and tractography were used to analyze white matter (WM) development in 11 newborns (age since conception <45 weeks) whose mothers were recruited during pregnancy. Comparisons were made with nine age-matched controls born to abstainers or light drinkers from the same Cape Coloured (mixed ancestry) community near Cape Town, South Africa. DTI parameters, T1 relaxation time, proton density and volumes were used to quantify and investigate group differences in WM in the newborn brains. Probabilistic tractography was used to estimate and to delineate similar tract locations among the subjects for transcallosal pathways, cortico-spinal projection fibers, and cortico-cortical association fibers. In each of these WM networks, the axial diffusivity was the parameter that showed the strongest association with maternal drinking. The strongest relations were observed in medial and inferior WM, regions in which the myelination process typically begins. In contrast to studies of older individuals with PAE, fractional anisotropy did not exhibit a consistent and significant relation with alcohol exposure. To our knowledge, this is the first DTI-tractography study of prenatally alcohol exposed newborns. (Taylor PA, Jacobson SW, van der Kouwe A, Molteno CD, Chen G, Wintermark P, Alhamud A, Jacobson JL, and Meintjes EM. Human Brain Mapping. 2014 Sept 3 [Epub].)
Significance: This study reveals a potential cellular mechanism contributing to prenatal ethanol exposure-induced increase in addiction risk. It suggests that the enhanced excitatory synaptic strength of dopamine neurons in the ventral tegmental area (VTA) may underlie the increased addiction propensity associated with fetal alcohol spectrum disorders (FASD).
Prenatal ethanol exposure (PE) is one of the developmental factors leading to increased addiction propensity (risk). However, the neuronal mechanisms underlying this effect remain unknown. We examined whether increased excitatory synaptic transmission in ventral tegmental area (VTA) dopamine (DA) neurons, which is associated with drug addiction, is impacted by PE. Pregnant rats were exposed to ethanol (0 or 6 g/kg/day) via intragastric intubation from gestational day 8-20. Amphetamine self-administration, whole-cell recordings, and electron microscopy were performed in male offspring between 2-12-week-old. The results showed enhanced amphetamine self-administration in PE animals. In PE animals, we observed a persistent augmentation in calcium-permeable AMPA receptor (CP-AMPAR) expression, indicated by increased rectification and reduced decay time of AMPAR-mediated excitatory postsynaptic currents (AMPAR-EPSCs), enhanced depression of AMPAR-EPSCs by NASPM (a selective CP-AMPAR antagonist), and increased GluA3 subunits in VTA DA neuron dendrites. Increased CP-AMPAR expression in PE animals led to increased excitatory synaptic strength and the induction of CP-AMPAR-dependent long-term potentiation (LTP), an anti-Hebbian form of LTP. These observations suggest that, in PE animals, increased excitatory synaptic strength in VTA DA neurons might be susceptible to further strengthening even in the absence of impulse flow. The PE-induced persistent increase in CP-AMPAR expression, the resulting enhancement in excitatory synaptic strength, and CP-AMPAR-dependent LTP are similar to effects observed after repeated exposure to drugs of abuse, conditions known to increase addiction risk. Therefore, these mechanisms could be important neuronal substrates underlying PE-induced enhanced amphetamine self-administration and increased addiction risk in individuals with fetal alcohol spectrum disorders. (Hausknecht K, Haj-Dahmane S, Shen YL, Vezina P, Dlugos C, and Shen RY. Neuropsychopharmacology. 2014 Oct 6 [Epub.]
Significance: A causal link between alcohol-induced microRNA (miRNA) expression and regulation of brain-derived neurotrophic factor (BDNF) on binge alcohol consumption was demonstrated by genetically manipulating the expression of the miRNA in the prefrontal cortex. These effects were specific to mice that transitioned from moderate to binge consumption. These results have implications for novel biomarker development to identify “problem drinkers” and suggest that novel pharmacotherapies targeting specific miRNAs may be beneficial in treating drinking problems in humans.
MicroRNAs (miRNAs) induce mRNA degradation and repress mRNA translation. Several miRNAs control the expression of the brain derived neurotrophic factor (BDNF) in the prefrontal cortex (PFC). The BDNF signaling pathway is activated by moderate intake of alcohol to prevent escalation to excessive drinking. Here, we present data to suggest that the transition from moderate to uncontrolled alcohol intake occurs, in part, upon a breakdown of this endogenous protective pathway via a miRNA-dependent mechanism. Specifically, a mouse paradigm that mimics binge alcohol drinking in humans produced a robust reduction in BDNF mRNA levels in the medial prefrontal cortex (mPFC), which was associated with increased expression of several miRNAs including miR-30a-5p. We show that miR-30a-5p binds the 3’ untranslated region (3’UTR) of BDNF, and that overexpression of miR-30a-5p in the mPFC decreased BDNF expression. Importantly, overexpression of miR-30a-5p in the mPFC produced an escalation of alcohol intake and a preference over water. Conversely, inhibition of miR-30a-5p in the mPFC using a Locked Nucleic Acid (LNA) sequence that targets miR-30a-5p restored BDNF levels and decreased excessive alcohol intake. Together, our results indicate that miR-30a-5p plays a key role in the transition from moderate to excessive alcohol intake. (Darcq E, Warnault V, Phamluong K, Besserer GM, Liu F, and Ron D. Mol Psychiatry. 2014 November 11 [Epub].)
Significance: This study shows that adolescent voluntary binge drinking reduces the density of myelinated axons in the medial prefrontal cortex and has long-lasting effects on prefrontal white matter. The significance of this finding is that it establishes a causal role for alcohol in the reduced density of myelin tracks and opens the door for more mechanistic studies.
Teen binge drinking is associated with low frontal white matter integrity and increased risk of alcoholism in adulthood. This neuropathology may result from alcohol exposure or reflect a pre-existing condition in people prone to addiction. Here we used rodent models with documented clinical relevance to adolescent binge drinking and alcoholism in humans to test whether alcohol damages myelinated axons of the prefrontal cortex. In Experiment 1, outbred male Wistar rats self-administered sweetened alcohol or sweetened water intermittently for 2 weeks during early adolescence. In adulthood, drinking behavior was tested under nondependent conditions or after dependence induced by 1 month of alcohol vapor intoxication/withdrawal cycles, and prefrontal myelin was examined 1 month into abstinence. Adolescent binge drinking or adult dependence induction reduced the size of the anterior branches of the corpus callosum, i.e., forceps minor (CCFM), and this neuropathology correlated with higher relapse-like drinking in adulthood. Degraded myelin basic protein in the gray matter medial to the CCFM of binge rats indicated myelin was damaged on axons in the mPFC. In follow-up studies we found that binge drinking reduced myelin density in the mPFC in adolescent rats (Experiment 2) and heavier drinking predicted worse performance on the T-maze working memory task in adulthood (Experiment 3). These findings establish a causal role of voluntary alcohol on myelin and give insight into specific prefrontal axons that are both sensitive to alcohol and could contribute to the behavioral and cognitive impairments associated with early onset drinking and alcoholism. (Vargas WM, Bengston L, Gilpin NW, Whitcomb BW, and Richardson HN. J. Neuroscience. 34(44):14777-14782; 2014.)
Significance: These findings show that the low expressing 5-HTTLPR genotype is associated with greater externalizing behavior among youth in substance use treatment, and that 5-HTTLPR genotype is associated with treatment outcome indirectly, through externalizing behavior. These findings suggest that youth with the low expressing 5-HTTLPR genotype and externalizing behaviors may benefit from interventions that address serotonergic function, and intensive treatment that simultaneously aims to reduce externalizing behaviors and substance use.
Meta-analyses suggest that the serotonin transporter linked polymorphic region (5-HTTLPR) short (S) allele, relative to the long (L) allele, is associated with risk for alcohol dependence, particularly among individuals with early onset antisocial alcoholism. Youth in substance use treatment tend to show antisocial or externalizing behaviors, such as conduct problems, which predict worse treatment outcome. This study examined a pathway in which 5-HTTLPR genotype is associated with externalizing behavior, and the intermediate phenotype of externalizing behavior serves as a link between 5-HTTLPR genotype and substance use treatment outcome in youth. Adolescents (n = 142) who were recruited from addictions treatment were genotyped for 5-HTTLPR polymorphisms (S and LG carriers vs. LALA), assessed for externalizing and internalizing behaviors shortly after starting treatment, and followed over 6-months. 5-HTTLPR genotype was not associated with internalizing behaviors, and was not directly associated with 6-month substance use outcomes. However, 5-HTTLPR genotype was associated with externalizing behaviors (S and LG > LALA), and externalizing behaviors predicted alcohol and marijuana problem severity at 6-month follow-up. Results indicated an indirect (p < 0.05) and non-specific (i.e., both alcohol and marijuana severity) effect of 5-HTTLPR genotype on youth substance use treatment outcomes, with externalizing behaviors as an important linking factor. Adolescents in substance use treatment with low expressing (S and LG) 5-HTTLPR alleles and externalizing behavior might benefit from intervention that addresses serotonergic functioning, externalizing behaviors, and substance use to improve outcomes. (Chung T, Cornelius JR, Martin CS, Ferrell R, Maisto SA, and Clark DB. Front Pediatr. 2014 Jul 7;2(71):1-7.)
Durable fear memories require PSD-95
Significance: Alcohol use disorders (AUDs) and trauma-related conditions such as post-traumatic stress disorder (PTSD) are highly co-morbid, but the pathological mechanisms underlying this relationship remain unclear. Genetically interfering with a key synaptic plasticity molecule, postsynaptic density 95 (PSD-95), in the prefrontal cortex led to a significant weakening of fear memories in mice. Targeting PSD-95-related mechanisms represents a novel approach to alleviating trauma-related anxiety associated with AUDs.
Traumatic fear memories are highly durable but also dynamic, undergoing repeated reactivation and rehearsal over time. Although overly persistent fear memories underlie anxiety disorders, such as posttraumatic stress disorder, the key neural and molecular mechanisms underlying fear memory durability remain unclear. Postsynaptic density 95 (PSD-95) is a synaptic protein regulating glutamate receptor anchoring, synaptic stability and certain types of memory. Using a loss-of-function mutant mouse lacking the guanylate kinase domain of PSD-95 (PSD-95GK), we analyzed the contribution of PSD-95 to fear memory formation and retrieval, and sought to identify the neural basis of PSD-95-mediated memory maintenance using ex vivo immediate-early gene mapping, in vivo neuronal recordings and viral-mediated knockdown (KD) approaches. We show that PSD-95 is dispensable for the formation and expression of recent fear memories, but essential for the formation of precise and flexible fear memories and for the maintenance of memories at remote time points. The failure of PSD-95GK mice to retrieve remote cued fear memory was associated with hypoactivation of the infralimbic (IL) cortex (but not the anterior cingulate cortex (ACC) or prelimbic cortex), reduced IL single-unit firing and bursting, and attenuated IL gamma and theta oscillations. Adeno-associated virus-mediated PSD-95 KD in the IL, not ACC, was sufficient to impair recent fear extinction and remote fear memory, and remodel IL dendritic spines. Collectively, these data identify PSD-95 in the IL as a critical mechanism supporting the durability of fear memories over time. These preclinical findings have implications for developing novel approaches to treating trauma-based anxiety disorders that target the weakening of overly persistent fear memories. (Fitzgerald PJ, Pinard CR, Camp MC, Feyder M, Sah A, Bergstrom H, Graybeal C, Liu Y, Schlüter O, Grant SGN, Singewald N, Xu W, and Holmes A. Molecular Psychiatry. 2014 Dec 16; 1-12.)
Significance: This study found that risk of injury due to excessive drinking differed between men and women. Risk of injury was similar for men and women up to three drinks; however, above three drinks, risk increased more rapidly for women than for men, and was significantly higher for women starting from 20 drinks. This suggests that alcohol attributable fractions for injury morbidity should be computed by gender.
AIMS: To update and extend analysis of the dose-response relationship of injury and drinking by demographic and injury subgroups and country-level drinking pattern, and examine the validity and efficiency of the fractional polynomial approach to modeling this relationship. DESIGN: Pair-matched case-cross-over analysis of drinking prior to injury, using categorical step-function and fractional polynomial analysis. SETTING: Thirty-seven emergency departments (EDs) across 18 countries. PARTICIPANTS: A total of 13 119 injured drinkers arriving at the ED within 6 hours of the event. MEASUREMENTS: The dose-response relationship was analyzed by gender, age, cause of injury (traffic, violence, fall, other) and country detrimental drinking pattern (DDP). FINDINGS: Estimated risks were similar between the two analytical methods, with injury risk doubling at one drink [odds ratio (OR) = 2.3-2.7] and peaking at about 30 drinks. Although risk was similar for males and females up to three drinks (OR = 4.6), it appeared to increase more rapidly for females and was significantly higher starting from 20 drinks [female OR = 28.6; confidence interval (CI) = 16.8, 48.9; male OR = 12.8; CI = 10.1, 16.3]. No significant differences were found across age groups. Risk was significantly higher for violence-related injury than for other causes across the volume range. Risk was also higher at all volumes for DDP-3 compared with DDP-2 countries. CONCLUSIONS: There is an increasing risk relationship between alcohol and injury but risk is not uniform across gender, cause of injury or country drinking pattern. The fractional polynomial approach is a valid and efficient approach for modeling the alcohol injury risk relationship. (Cherpitel CJ, Ye Y, Bond J, Borges G, and Monteiro M. Addiction. 2014 Oct 30 [Epub].)
Osteopontin deficiency does not prevent but promotes alcoholic neutrophilic hepatitis in mice
Significance: This paper advances a potential solution to a long-standing challenge for the study of human alcoholic liver disease, namely the lack of animal models with pathological end points and severity comparable to those observed in human patients. The Tsukamoto laboratory, building on the “binge on chronic” model used by others in the field, report hepatocyte necrosis and neutrophil infiltration, characteristic features of human alcoholic steatohepatitis not previously reproduced in animal models. Combining a diet enriched with saturated fat and cholesterol and continual intra-gastric alcohol infusion, plus a weekly “binge” administration of alcohol via gavage, generates a high level of liver injury in mice including histopathological and some clinical features (including elevated serum bilirubin) remarkably similar to those observed in human patients. Using this approach, the authors further demonstrated that osteopontin exerts a significant protective role. This new animal model is a highly promising tool for better understanding of human disease mechanisms and new intervention development.
Alcoholic hepatitis (AH) is a distinct spectrum of alcoholic liver disease (ALD) with intense neutrophilic (PMN) inflammation and high mortality. Although a recent study implicates osteopontin (SPP1) in AH, SPP1 is also shown to have protective effects on experimental ALD. To address this unsettled question, we examined the effects of SPP1 deficiency in male mice given 40% calories derived from ad libitum consumption of the Western diet high in cholesterol and saturated fat (HCFD) and the rest from intragastric feeding (iG) of alcohol diet without or with weekly alcohol binge. Weekly binge in this new hybrid feeding model shifts chronic ASH with macrophage inflammation and perisinusoidal and pericelluar fibrosis to AH in 57% (15/26) of the mice, accompanied by inductions of chemokines (Spp1, Cxcl1, Il-17a), progenitor genes (Cd133, Cd24, Nanog, Epcam), PMN infiltration, and clinical features of AH such as hypoalbuminemia, bilirubinemia, and splenomegaly. SPP1 deficiency does not reduce the AH incidence and inductions of progenitor and fibrogenic genes but rather enhances the Il-17a induction and PMN infiltration in some mice. Further, in the absence of SPP1, chronic ASH mice without weekly binge begin to develop AH. In conclusion, these results suggest SPP1 has a protective rather than causal role for experimental AH reproduced in our model. (Lazaro R, Wu R, Lee S, Zhu NL, Chen CL, French SW, Xu J, Machida K and Tsukamoto H. Hepatology. 2014 Nov 25 [Epub].)
NIAAA COMMUNICATIONS & MEDIA COVERAGE
Recent News Media Interviews
Dr. Koob gave numerous interviews to representatives of national and international news media on a number of timely topics related to NIAAA’s research and its impact on treatment and prevention of alcohol abuse and alcohol use disorders. These include interviews with The Wall Street Journal, Chicago Tribune,Houston Chronicle, National Public Radio among others. Dr. Koob also continues to serve as an advisor to HBO producers who are developing a documentary on alcohol abuse.
NIAAA division directors and other NIAAA experts were also frequent sources of information for reporters from a broad range of domestic and international news organizations.
FASD Awareness Day
On September 9, 2014, NIAAA recognized FASD Awareness Day. Outreach efforts were detailed in the September Director’s Report. A subsequent analysis of the Twitter chat featuring Dr. Ken Warren indicated that messages from the chat reached a potential 721,000 Twitter users. In addition, Parents Magazine (Parents.com) created and posted a video based on NIAAA messages.

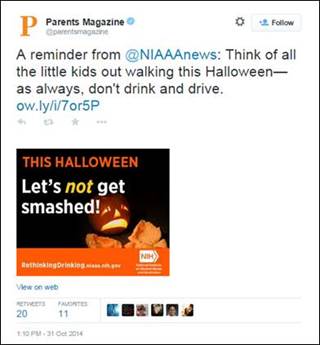
Seasonal Outreach - Halloween Safety
During October 2014, NIAAA’s Halloween safety graphic messages were tweeted and/or posted on Facebook by Parents Magazine, Working Mother Magazine, Military Spouse Magazine, Mothers Against Drunk Driving, and Choices Magazine (Scholastic), as well as several NIAAA Liaison Group members (including the American Society of Addiction Medicine and the American Pharmacists Association) and various bloggers. People.com, with more than 10 million unique visitors each month, ran both images in its advertising boxes at no charge.
Release and Promotion of New Booklet on Treatment Options for Alcohol Problems
During November 2014, NIAAA released a new publication titled “Treatment for Alcohol Problems: Finding and Getting Help.” This booklet, available in print and at: https://pubs.niaaa.nih.gov/publications/Treatment/treatment.pdf, covers the latest research-based treatments and what to consider when choosing among them.
Promotional efforts have encompassed social media, stakeholder outreach, and pitching to selected news media outlets and science writers. Thus far, these efforts have resulted in a Wall Street Journal article, a 4-page feature appearing in the February 2015 issue of the National Library of Medicine’s Medline Plus magazine, and an article in an upcoming issue of NIH News in Health. Several liaison groups, including the American Psychiatric Association and American Society of Addiction Medicine, have informed their members about the publication through their newsletters.
Winter Holiday Outreach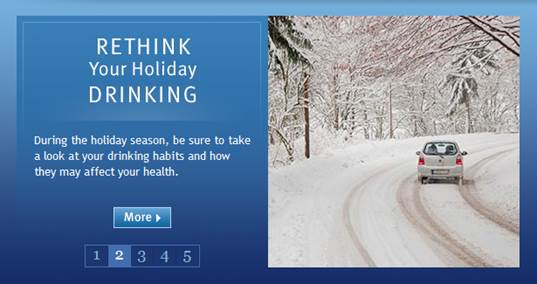
During December 2014, NIAAA continued its seasonally-focused outreach through dissemination of the holiday fact sheet to news media, community organizations, and local police and highway departments. In addition, NIH ran the NIAAA graphic pictured here on the main home page.
Communications and Public Liaison Branch (CPLB) Outreach – New Liaison Group Activities
Since September, CPLB has initiated new collaborative projects with a number of new and existing liaison groups:
- APA (American Psychiatric Association) – newsletter article promoting treatment publication, Twitter chats, sharing of social media graphics
- ASAM (American Society of Addiction Medicine) – newsletter article
- NCADD (National Council on Alcoholism and Drug Dependence) – potential collaborations including Twitter chats, based on discussions with new Executive Director Andrew Pucher
- Righttime Urgent Care – images with NIAAA logo and messaging for Halloween, Holiday/New Year’s Eve drinking, and summer drinking safety on display in patient waiting rooms at their 13 locations throughout Maryland
- Community of Concern – supporting translation and printing of NIAAA’s treatment booklet and creating a special run of the booklet with NIAAA messages
- In addition, NIAAA has participated in several recent Twitter chats, including a SAMHSA-sponsored chat on underage drinking, and a discussion of the recent Monitoring the Future findings with NIDA.
Publication Statistics
As of the end of November, there were 28,879 subscribers to NIAAA’s Alcohol Alert; 27,912 to Alcohol Research: Current Reviews; 18,306 to the NIAAA Spectrum; and 18,038 to receive general information. NIAAA also reached a social media milestone in November – we now have more than 10,000 followers on Twitter.

